FDA warns of CARDIAC INFLAMMATION risk linked to the Novavax COVID-19 vaccine
06/05/2022 / By Ramon Tomey
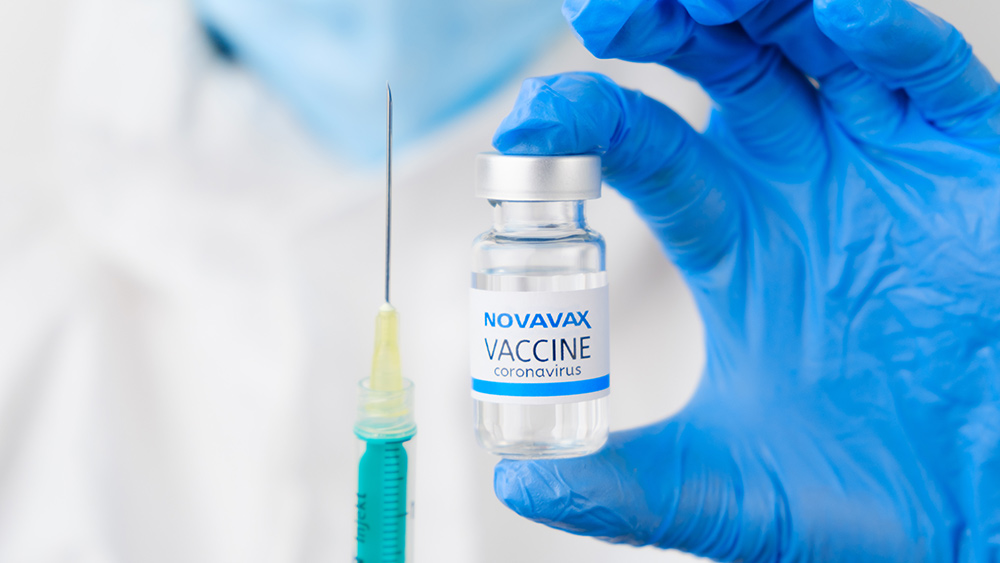
Food and Drug Administration (FDA) staffers warned of the potential cardiac inflammation risk linked to the Wuhan coronavirus (COVID-19) vaccine from Novavax.
The regulatory agency’s staff members pointed out the risk in documents released June 3, ahead of the FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) meeting scheduled on June 7. They cited four events of myocarditis – inflammation of the heart muscle – that occurred during clinical trials of the vaccine. The Maryland-based Novavax conducted trials of its COVID-19 vaccine on almost 30,000 patients between December 2020 and September 2021.
“These events raise the concern for a causal association with this vaccine, similar to the association documents with mRNA COVID-19 vaccines. Post-marketing data from individuals receiving mRNA vaccines [has] demonstrated increased risks of myocarditis and pericarditis, particularly within seven days following the second primary dose,” the FDA staffers noted. Pericarditis pertains to the inflammation of the pericardium or the lining surrounding the heart. (Related: Heart inflammation reported in teens following Moderna and Pfizer vaccine.)
“Data from passive surveillance during post-authorization use in other countries also indicate a higher than expected rate of myocarditis and pericarditis … associated with the vaccine. Further evaluation is needed to inform the risk of myocarditis and pericarditis, and their outcomes, as additional data emerge over time.”
Aside from the cardiac inflammation risks, the FDA staffers also cited tenderness at the injection site, headache, fatigue and muscle pain as immediate reactions. They also mentioned hypersensitivity and swelling of the lymph nodes as reactions linked to the Novavax shot. There was also one case of Guillain-Barre syndrome – which is “known to be associated with other [COVID-19] vaccines” – observed during the Novavax clinical trial.
Novavax’s COVID-19 vaccine is different from the other vaccines in the market, as it uses an insect virus to deliver the SARS-CoV-2 spike protein. The Pfizer and Moderna mRNA vaccines use lipid nanoparticles containing messenger RNA to deliver the spike protein in the body. Meanwhile, the AstraZeneca and Janssen viral vector vaccines use an adenovirus for this purpose.
The Novavax vaccine also contains the compound saponin, which is taken from the soap bark tree (Quillaja saponaria) native to South America. Saponin’s soap-like qualities trigger a response from the body’s immune system when injected in the body.
Novavax shot causes catastrophic reactions in three Aussie women
The U.S. has not yet authorized the Novavax shot as of writing – but some countries including the Philippines, the United Kingdom, Indonesia, India, South Korea and Australia have approved it. The Therapeutic Goods Administration, the Australian counterpart of the FDA, authorized the Novavax vaccine in January 2022. It recommended the two-dose vaccine for Australians aged 18 and older, with a three-week interval between both.
However, The COVID Blog outlined three cases of women suffering from serious vaccine injuries after being injected with the shot.
A 42-year-old woman named Tasha received her first Novavax dose on Feb. 21. She immediately reported a “hot gushing feeling down [her] throat that tasted metallic,” a “deep throbbing headache,” profuse sweating and “the worst chest pain.” The resident of Adelaide, South Australia sought medical treatment only to be told that doctors did not know how to deal with the adverse reactions caused by the Novavax vaccine. Tasha became completely bedridden by March 1.
Lee-Anne Barnett, 45, also reported the same metallic taste immediately after getting her first Novavax dose on Feb. 18. Following several days of having a sore arm, the Adelaide resident developed very itchy rashes on both thighs that rapidly spread to her legs. Doctors only prescribed steroids for the rashes, dismissing the possibility that the COVID-19 vaccine had something to do with it. Barnett reported “diminishing cognitive abilities” that made her feel like she was “getting dementia” on March 2.
Chiara Clowes, 30, experienced a plethora of reactions – including difficulty breathing, extreme chest pain, extreme fatigue, brain fog and cognitive issues – after getting the vaccine on Feb. 17. While tests conducted by medical professionals found nothing, they acknowledged Clowes’ chest pain was caused by the Novavax shot. On Feb. 24, doctors found a cyst in her ovary. Clowes, who lives a healthy lifestyle prior to her COVID-19 vaccination, now has to regularly meet with gynecologists as a consequence.
“What we know for certain is that Novavax is an experimental injection and is already making people sick,” remarked The COVID Blog.
Watch Dr. Jane Ruby expounding on the dangers of the Novavax COVID-19 vaccine below.
This video is from the Information Warfare channel on Brighteon.com.
More related stories:
3 Aussie women experience SERIOUS REACTIONS after getting Novavax COVID shot.
Operation Warp Speed gives $1.6 billion in taxpayer funding to Novavax for coronavirus vaccines.
Sources include:
Submit a correction >>
Tagged Under:
Big Pharma, cardiac inflammation, covid-19, FDA, heart health, heart inflammation, myocarditis, Novavax, pandemic, pericarditis, risk warning, serious reactions, vaccine, vaccine injury, vaccines, Wuhan coronavirus
This article may contain statements that reflect the opinion of the author
RECENT NEWS & ARTICLES
Pandemic.News is a fact-based public education website published by Pandemic News Features, LLC.
All content copyright © 2018 by Pandemic News Features, LLC.
Contact Us with Tips or Corrections
All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.